Agilis Upcoming Events
Lets connect at one of our upcoming events!

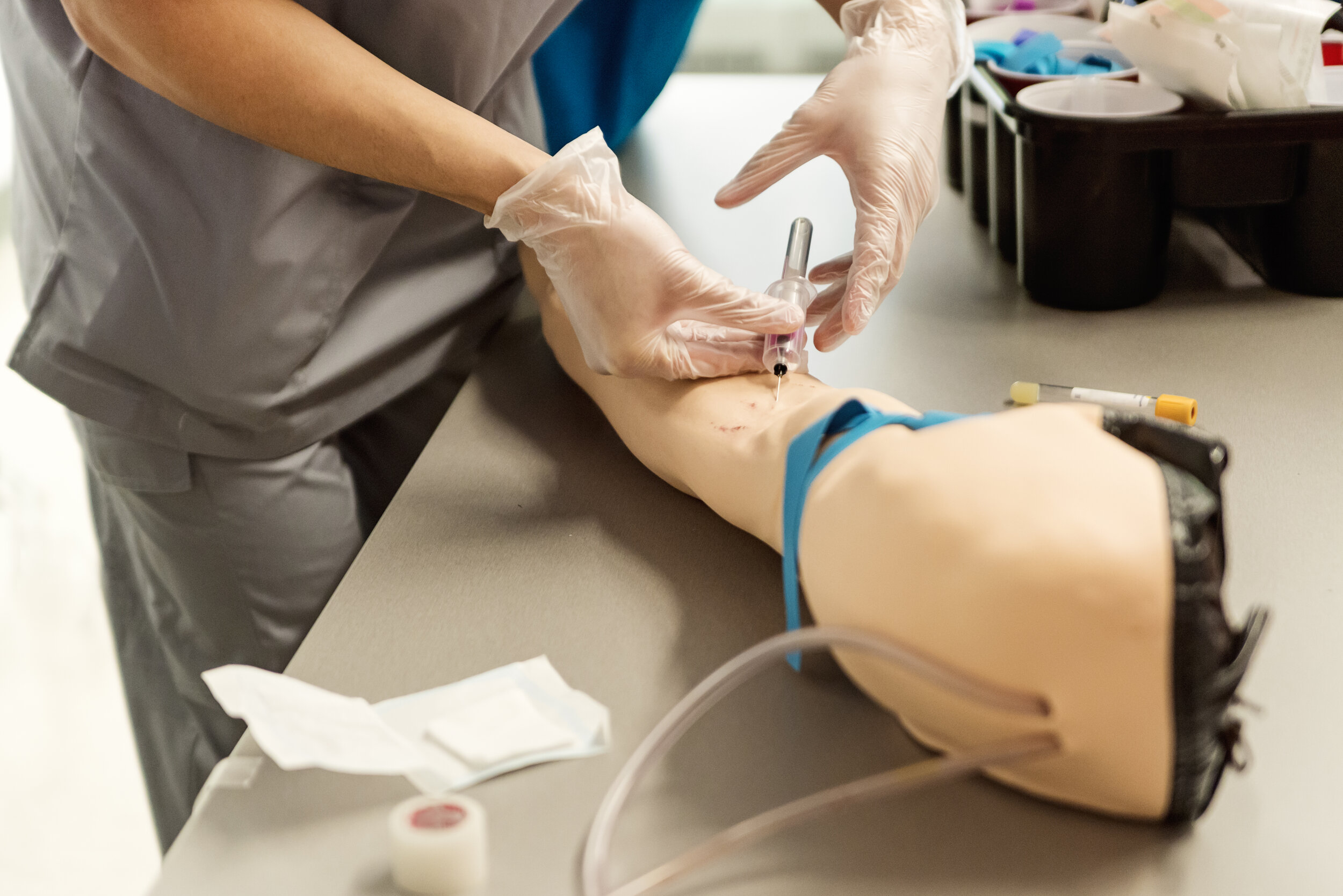
Agilis has global experience in planning your next Human Factors study

Who we are
Agilis by Kymanox is your trusted human factors partner and end-to-end solutions provider for the life sciences industry. Our mission is to help our clients provide end users with safer and more reliable medical devices and we are proud of our success rate!
Choosing Agilis as your human factors partner provides the advantage you need to navigate the complex global regulatory landscape and achieve successful human factors submission the first time!
What we do
For the last 20 years we have helped new and established companies achieve successful human factors submissions while navigating the complex global regulatory landscape, resulting in safe and effective medical devices and combination products.
Together we can provide human factors, regulatory, and usability engineering team to support our clients programs from start through regulatory submissions and commercialization
Why we do it
Agilis by Kymanox values our right-first-time submissions, uncompromising quality, and always a focus on the end user and patient.
We work with our partners to successfully navigate the human factors engineering design process.
Our Advantage
FDA has in recent years started tracking human factors submission success, and reports only 4-11% of HF submissions are accepted upon the first submission with no requests for additional information.
Our clients count on us to understand their specific regulatory environments and provide experienced guidance to help ensure the fastest and most complete studies and reports. We customize our services to meet each client’s needs and specific challenges

Global Experience
20+
Years in Business
Our clients count on us to understand their specific regulatory environments and provide experienced guidance to help ensure the fastest and most complete studies and reports. We customize our services to meet each client’s needs and specific challenges.
75+
Years Combined Experience
Agilis’ team are all seasoned human factors experts, many of whom are considered industry thought leaders. We strive to maintain consistency of your Agilis team throughout the project lifecycle. Our unabashed commitment to quality is evidenced by a rigorous internal quality process including on-going professional development for all Agilis team members.

Stay Connected
Read our News
Kymanox’s acquires Agilis. Agilis by Kymanox is still your trusted human factors partner and now an end to end solutions provider for the life sciences industry.
Visit us at Events
We would like the chance to interact with you throughout the year. Stay up to date with events where Agilis is participating or supports.
Experience
Our work is exclusive in the life sciences industry and we have experience with various types of regulatory submissions and devices.